When a child has ongoing stomach pain, diarrhea, or other unexplained symptoms, the typical workup includes blood panels, traditional stool tests, and imaging. Each takes time, adds cost, and may raise more questions than it answers. If results aren’t clear, the next step is often invasive: colonoscopy or endoscopy, with sedation, infection risk, and bills that can reach thousands.
Dr. Qian Yuan, a pediatric gastroenterologist at Massachusetts General Hospital and Associate Professor at Harvard Medical School, knows this process well. As a Tiny Health Medical Advisor, he also contributes to microbiome research, reshaping what pediatric diagnostics can look like.
Through Powered by Tiny, gut microbiome testing is now part of Dr. Yuan's workup from day one, whether on its own or alongside standard tests. For him, the question isn't whether gut testing belongs in clinical care, but how quickly others will adopt it.
He’s among a growing number of clinicians who are bringing microbiome testing into their practices. Sutter Health, Kaiser Permanente, Mayo Clinic, Allina Health, MNGI, and UCI Health have also adopted Tiny Health gut microbiome testing.
Dr. Yuan’s experience at one of the country's most respected academic medical centers offers an inside look at what this protocol can mean for patients, clinical workflows, and insurers.
Where standard diagnostics fall short
Dr. Yuan's patients include children with chronic stomach pain, constipation, diarrhea, recurrent SIBO (small intestinal bacterial overgrowth), reflux, and complex immune-related food conditions. Before adding microbiome testing, his workup followed standard pediatric GI protocols: blood panels, conventional stool tests for inflammation, digestive markers, and pathogen detection via qPCR, SIBO breath testing, abdominal ultrasounds, and, when needed, endoscopy or colonoscopy.
Even when used conservatively, roughly 30% of those scopes came back negative or inconclusive. It's a key reason Dr. Yuan makes Tiny Health testing the first step, not the last.
Colonoscopies and upper endoscopies cost insurers a national average of $5,100–$6,250 [1]. Those figures include pathology, office visits, and anesthesia (full sedation is always required in pediatric cases). For a health plan covering 1,000 similar pediatric GI patients each year, that 30% rate translates to $700,000–$900,000 in potentially avoidable spend.
When those procedures offer no answers, what follows is more waiting, more appointments, and more tests. The diagnostic clock keeps running, and so does the bill.
Microbiome testing helps address these gaps. It delivers microbial data early, often before imaging or scopes are needed.
A different entry point
Dr. Yuan began using Tiny Health gut microbiome testing as an early screening tool, a shift toward finding actionable data before taking invasive steps.
For infants with suspected FPIAP (food protein-induced allergic proctocolitis) or FPIES (food protein-induced enterocolitis syndrome), he now runs a Tiny Health microbiome analysis before ordering other tests. With older children, Dr. Yuan pairs it with blood work and a traditional qPCR stool test. qPCR panels test for a narrow set of known pathogens, so when results come back inconclusive, Tiny Health fills in the clinical picture and helps shorten the diagnostic path.
When the gut microbiome is out of balance (dysbiosis), the downstream effects can look like almost anything: chronic pain, irregular stool patterns, reflux, recurring infections. Microbiome sequencing can sometimes reveal microbial drivers behind symptoms that standard labs may miss.
"If we choose Tiny Health as the first-line workup," Dr. Yuan says, "in some patients, we probably can avoid invasive procedures such as EGD and colonoscopy, or imaging and other tests."
The cost of diagnostic uncertainty: one patient’s path
A 16-year-old patient saw Dr. Yuan after extensive testing elsewhere. His prior workup included qPCR stool panels, a SIBO breath test, and multiple blood panels. When none of those provided answers, the diagnostic path progressed to capsule endoscopy and MR enterography, and ultimately an upper endoscopy and colonoscopy under general anesthesia.
Those tests alone cost $13,200; specialist visits added thousands more. Still, his results were inconclusive.
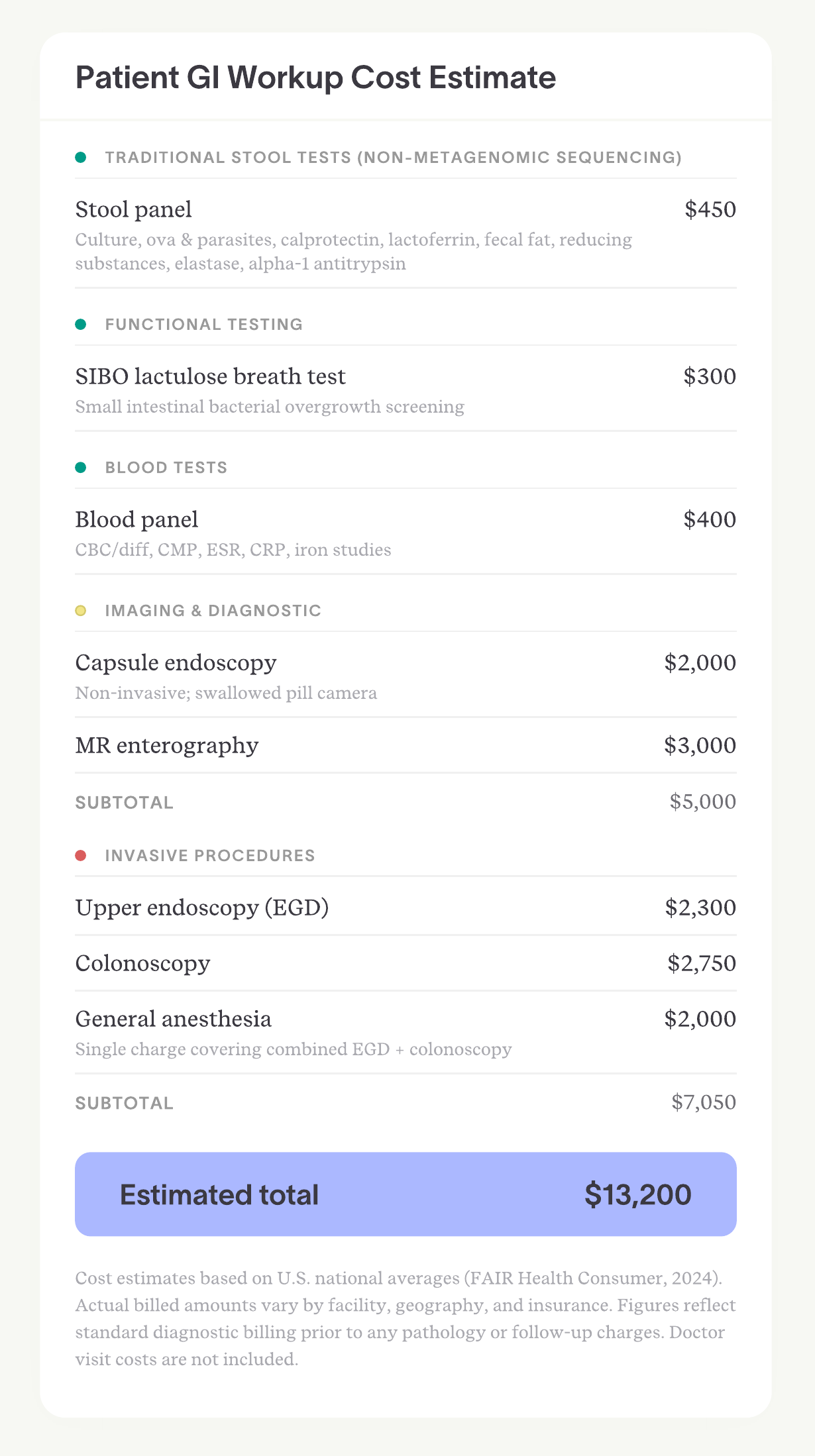
Dr. Yuan ordered a Tiny Health gut test, which pinpointed microbial imbalances linked to symptoms. This guided effective treatment.
For this teenager, his family, and their insurer, this case shows what's possible when microbiome testing leads rather than follows.
A colonoscopy costs 13x more than Tiny Health testing
A Tiny Health gut microbiome test is $199 for healthcare providers. An inconclusive colonoscopy costs insurers about 13 times more.
Screening tests that guide treatment protocols earlier, or help avoid procedures, can mean real savings for patients and insurers.
"Tiny Health results provide direct evidence of a patient's gut microbiome composition and functions," Dr. Yuan explains, "and by correcting those imbalances, we can improve quality of life and avoid other expensive, invasive testing."
What this means for insurance companies and payers
The financial case for first-line microbiome testing is real. Every inconclusive colonoscopy or endoscopy means payment for a procedure that didn’t help the patient. More imaging, labs, and specialist referrals follow, each adding cost.
A $199 microbiome test that provides answers early in a workup can cut downstream spending. For payers covering claims where invasive procedures require sedation, the savings potential is even greater.
Reimbursement decisions require documentation. Dr. Yuan summarizes Tiny Health findings in his clinical notes to referring primary care physicians, creating a record that links microbiome data to care decisions. Tiny Health aims to provide a short summary clinicians can add directly to their notes, making it easy for payers to see how microbiome testing informs or, in some cases, replaces costlier steps. That kind of structured documentation is what builds the evidence base reimbursement policy requires.
As more GI clinicians use shotgun metagenomics and document microbiome-informed care, the cumulative data will strengthen the case for reimbursement and broader clinical adoption.
Savings per patient are hard to ignore. Across this complex pediatric specialty, the impact is significant.
The case for wider adoption
Dr. Yuan is working to build awareness at Mass General, though he hasn't had formal discussions with his GI colleagues about microbiome testing. He expects the barrier to be familiarity, not skepticism.
"It all depends on the doctor's knowledge of the gut microbiome and guiding families based on the results," he says.
Clinicians confident in interpreting microbiome data and translating it into actionable guidance will adopt it. Tiny Health offers education resources and one-on-one provider onboarding to support that learning curve. Peer exposure and real-world results like Dr. Yuan's may help close the remaining gap.
Patient demand may speed adoption. Parents of Dr. Yuan's patients often express interest in gut testing. "They want me to order Tiny Health kits for everyone in the family," he says.
The way forward
“Tiny Health is the gut microbiome test I've long wished for at Mass General. Now that it's here, it gives me evidence-based data to guide my patients’ treatment,” Dr. Yuan says.
He's not alone. Across leading academic medical centers and health systems, clinicians are increasingly integrating microbiome testing into standard GI care.
"By equipping providers with comprehensive microbiome testing, our aim is to help shift GI care toward a more preventive, personalized model, whether in pediatric or adult clinics," says Tiny Health founder Cheryl Sew Hoy.
Learn the root cause of your patients’ symptoms faster and improve health outcomes using Powered by Tiny in your practice.